MP Board Class 12th Chemistry Important Questions Chapter 11 Alcohols, Phenols and Ethers
Alcohols, Phenols and Ethers Important Questions
Alcohols, Phenols and Ethers Very Short Answer Type Questions
Question 1.
What is Ether?
Answer:
Compounds formed by the substitution of hydrogen atom of hydrocarbon by alkoxy group are called Ethers.
Question 2.
What will be the type of alcohol formed by the hydration of propene in the presence of acid?
Answer:

Question 3.
What is the special name of Phenol and from what was it first isolated?
Answer:
Phenol is also known as carbolic and it was first isolated from coal tar.
![]()
Question 4.
Picric acid is a strong acid. Why?
Answer:
In picric acid, acidic character increases due to the presence of three electron attracting – NO2 groups because these groups are helpful in the release of H+. Thus picric acid is a strong acid.
Question 5.
Write the reagent required for the preparation of tertiary butyl alcohol starting from propanone.
Answer:
Methyl magnesium bromide.
Question 6.
What type of isomerism is exhibited between alcohol and ether?
Answer:
Alcohol and ether exhibit Functional isomerism.
Question 7.
Write the equation of catalytic reduction of Butanols.
Answer:

Question 8.
Write IUPAC name of the following compound.

Answer:

IUPAC name of this compound is 2,2, 3 – trimethyl pentan – 1 – ol.
Question 9.
Why do ethers have low boiling points?
Answer:
Molecules of ether do not possess H – bonding, therefore boiling points of ethers are low.
Alcohols, Phenols and Ethers Short Answer Type Questions
Question 1.
What is Lucas reagent? How are primary, secondary and tertiary alcohol identified by it? Explain.
Answer:
Mixture of anhydrous ZnCl2 and cone. HCl is known as Lucas reagent.
1. Tertiary Alcohol : On adding Lucas reagent in alcohol at normal temperature, immediately white oily precipitate of Alkyl chlorides is formed, then it is tertiary alcohol.
2. Secondary Alcohol : If on adding Lucas reagent in alcohol, at normal temperature, a white oily precipitate of alkyl chloride is obtained after 5 minutes, then it is secondary alcohol.
3. Primary Alcohol : Primary alcohol does not show any reaction with Lucas reagent at normal temperature.
![]()
Question 2.
Why the b.p. of alcohol are higher than ethers and alkene?
Or C3H5OH and CH3OCH3 both have same molecular formula (C2H6O) but the b.p. of alcohol is 78.4°C and b.p. of ether is – 240°C. Explain the reason.
Answer:
In case of C2H5OH there is strong intermolecular hydrogen bonding between the molecules of alcohol. So alcohols (C2H5OH) required much energy to evaporate than ether molecules. In other words, we can say that the C2H5OH molecules are in associated form due to H – bonding so the b.p. of C2H5OH is very higher than ether and alkene.

Question 3.
Boiling point of alcohol higher than corresponding alkane. Why?
Answer:
Boiling point of alcohols is much higher than hydrocarbons of nearly similar molecular mass due to inter molecular hydrogen bond. Alcohols molecules associate kilo calories mole-1. Thus, extra energy is required for the separation of these molecules, which lead to increase in boiling point. Hydrocarbons do not form hydrogen bond, thus their boiling point is comparatively less.
Question 4.
- How can we obtain phenol from benzene diazonium chloride?
- What is the reaction of diethyl ether with HI acid?
Answer:
1. Phenols are prepared by hydrolysis of diazonium salts by water, dil. acids etc.

2. The reaction of diethyl ether with cone. HI acid, on heating gives one molecule of ethyl iodide and one molecule of ethyl alcohol.

Diethyl ether Ethyl iodide Ethyl alcohol
Question 5.
Write the equations involved in the following reactions :
- Reimer – Tiemann reaction (NCERT, MP2018)
- Kolbe’s reaction.
Answer:
1. Reimer – Tiemann reaction:
When phenol is treated with chloroform in presence of aqueous sodium hydroxide at 60°C, oHydroxy benzaldehyde (Salicylaldehyde) and p – Hydroxy benzaldehyde are formed. The ortho – isomer is the major product. This reaction is called Reimer-Tiemann reaction.

If carbon tetrachloride is used in place of chloroform, salicylic acid is obtained as the main product.

2. Kolbe – Schmidt reaction:
When sodium salt of a phenol is heated with CO2 at 130°C. (403K) and 4 – 7 atm pressure, sodium salicylate is formed. This on acidification gives salicylic acid.

At high temperature p – derivative is formed.

Question 6.
Name the reagents used in the following reactions: (NCERT)
- Oxidation of a primary alcohol to carboxylic acid.
- Oxidation of a primary alcohol to aldehyde.
- Bromination of phenol to 2,4,6 – tribromo – phenol.
- Benzyl alcohol to benzoic acid.
- Dehydration of propan – 2 – ol to propene.
- Butan – 2 – one to butan – 2 – ol.
Answer:
- Acidified K2Cr2O7 or neutral acidic or alkaline KMnO4.
- Pyridinium chlorochromate (pcc) in CH2Cl2 or Cu at 573K.
- Bromine water (Br2/H2O)
- Acidified or alkaline KMnO4
- Conc. H2SO4 at 443K or 85% phosphoric acid at 443K.
- Ni/H2 or NaBH4 or LiAlH4.
Question 7.
Explain, how does the – OH group attached to a carbon of benzene ring activate it towards electrophilic substitution? (NCERT)
Answer:
The – OH group exerts +R effect on the benzene ring under the effect of attacking electrophile. As a result, there is an increase in the electron density in the ring particularly at ortho and para positions, therefore electorphilic substitution occurs mainly at o – and p – positions.
![]()
Question 8.
Write Victor Meyer method to distinguish primary, secondary and tertiary alcohol.
Answer:
Victor Meyer’s method:
1. The given alcohol is converted into an iodide by concentrated HI or red phosphorus and iodine.
2. The iodide is treated with silver nitrite to form nitroalkane.
3. Nitroalkane is finally treated with nitrous acid (NaNO2 + H2SO4) and made alkaline with KOH.
- If a blood red colour is obtained, the original alcohol is primary.
- If a blue colour is obtained, the alcohol is secondary.
- If no colour is produced, the alcohol is tertiary.

Question 9.
Give the equations of reactions for the preparation of phenol from cumene.
Answer:
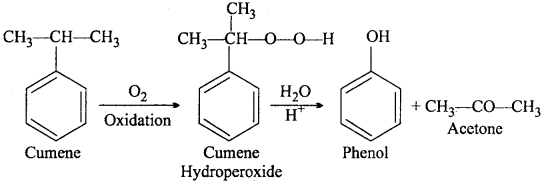
Question 10.
Differentiate between Phenol and Alcohol and write Libermann’s reaction related to phenol.
Answer:
Differences between Phenol and Alcohol:
Phenol:
- Physical properties – Characteristic phenolic odour, sparingly soluble in water.
- It is acidic and dissolve in bases to form salt.
- On oxidation, hybrid coloured product is formed.
- Produce characteristic colour with Ferric chloride.
- It does not react with halogen acid.
- With PC15, mainly form triaryl phosphate.
Alcohol:
- Pleasant odour, fairly soluble in water.
- It is neutral and do not reacts with bases.
- It can easily oxidize to Aldehydes and ketones.
- It does not reacts with ferric chloride.
- Forms Alkyl halide.
- Alkyl chloride are formed.
Libermann’s Reaction:
On adding few drops of concentrated sulphuric acid and little sodium nitrite in phenol first dark blue colour is produced on adding water colour becomes red and on adding an alkali red colour again changes to blue colour.
![]()
Question 11.
Give equations for the preparation of ethyl alcohol by starch and write name of enzymes.
Answer:
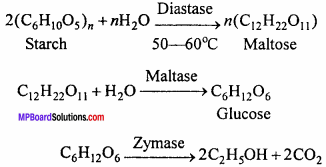
Enzymes:
- Diastase
- Maltase
- Zymase.
Question 12.
Explain, why propanol has higher boiling point than that of the hydrocarbon, butane? (NCERT)
Answer:
The molecules of butane are held together by weak van der Waals’ force of attraction while those of propanol are held together by stronger intermolecular hydrogen bonding.

Therefore, the b.p. of propanol is much higher than that of butane.
Question 13.
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact. (NCERT)
Answer:
Alcohols can form hydrogen bonds with water and break the H – bond exist between water molecules. Hence, they are soluble in water.

On the other hand, hydrocarbons cannot form hydrogen bonds with water molecules and hence are insoluble in water.
Question 14.
What is meant by hydroboration – oxidation reaction? Illustrate it with an example. (NCERT)
Answer:
The addition of diborane to alkene to form trialkyl boranes followed by their oxidation with alkaline hydrogen peroxide to form alcohol is called hydroboration – oxidation. For example:

The alcohols obtained by this process appear to have been formed by direct addition of water to the alkene against Markownikoffs rule.
Question 15.
Ethyl alcohol and phenol both contain – OH group. What is the reason that phenoffs acidic and alcohol has alkaline effect? (MP 2015)
Or
Ethyl alcohol and phenol both contain – OH group. What is the reason that phenol is acidic and alcohol is neutral in nature? (MP 2013)
Answer:
Explanation of acidic nature of phenol:
One possible explanation why phenols are stronger acids as compared to alcohols is that phenols exist as a resonance hybrid.

Due to resonance, the oxygen atom gets a positive charge and attracts the electron pair of the O – H bond and thus facilitates the release of a proton. The phenoxide ion formed after the release of a proton is also stabilized by resonance.
![]()
In alcohols, no resonance is possible hence the hydrogen atom is more firmly linked to the oxygen.
![]()
Question 16.
Pure phenol is a colourless solid but why it is converted into pink after some time?
Or
What change in colour is observed in phenol in presence of oxygen? Explain with reaction. (MP2011)
Answer:
In the presence of air pure phenol oxidises into quinone.

This quinone again combines with two molecules of phenol by H – bond and gives pink phenoquinone.

Question 17.
Explain the manufacture of CH3OH by water gas.
Answer:
From water gas : Steam is passed over red hot coke when water gas is formed.

Water gas is mixed with half its volume of hydrogen, compressed to about 200 atm and passed over a catalyst which is a mixture of oxides of copper, zinc and chromium at 300°C.

Alcohols, Phenols and Ethers Long Answer Type Questions
Question 1.
What is Williamson continuous etherification process? Is it a continuous process? Explain. Give labelled diagram.
Or,
Describe the laboratory method of preparation of diethyl ether. How ether thus obtained is purified?
Answer:
Laboratory Method for the Preparation of Diethyl Ether (Sulphuric Ether):
Diethyl ether is prepared in the laboratory and industry by the Williamson continuous etherification process, i.e., by heating ethanol (in excess) with concentrated sulphuric acid.

Sulphuric acid is regenerated in the reaction hence, it appears as if only a small amount of acid may convert an excess of alcohol into ether. So, this method is called Williamson continuous etherification process but actually we cannot get ether continuously.
This is due to the following two reasons :
- Water formed in the reaction dilutes the acid and its reactivity decreases.
- A part of sulphuric acid is reduced by alcohol into sulphur dioxide.
Method:
Ethanol and H2SO4 (2:1) are taken in a flask and heated on sand bath at 140°C. Ethanol is added at the same rate at which ether distilled over and is collected in a receiver cooled in ice – cold water.

Purification:
Ether contain ethanol, water and sulphuric acid as impurities. It is washed with NaOH to remove sulphuric acid and then agitated with 50% solution of calcium chloride to remove alcohol. It is then washed with water, dried over anhydrous calcium chloride and redistilled.
Question 2.
How can you change the following:
- Methanol to ethanol
- Ethanol to methanol.
Answer:
1. Methanol to ethanol :

2. Ethanol to methanol :

Question 3.
Differentiate primary, secondary and tertiary alcohol by oxidation and dehydrogenations method. (MP 2011)
Answer:
1. Oxidation:
The oxidizing agents generally used for oxidation of alcohols are acid dichromate, acid or alkaline KMnO4 and dilute HNO3.
(i) A primary alcohol is easily oxidized to an aldehyde and then to an acid both containing the same number of carbon atoms as the original alcohol.

(ii) A secondary alcohol on oxidation gives a ketone with the same number of carbon atoms as the original alcohol, ketones are oxidized with difficulty but prolonged action of oxidizing agents produce carboxylic acids containing fewer number of carbon atoms than the original alcohol.

(iii) A tertiary alcohol is resistant to oxidation in neutral or alkaline solutions but is readily oxidized by an acid oxidizing agent giving a mixture of ketone and acid each having lesser number of carbon atoms than the original alcohol.

2. Dehydrogenation (Action of hot reduced copper at 300 °C):
Different types of alcohols give different products when their vapours are’passed over Cu gauze at 300°C.
Primary alcohols lose hydrogen and yield an aldehyde.

Secondary alcohols lose hydrogen and yield a ketone.

Tertiary alcohols are not dehydrogenated but lose a water molecule to give alkenes.

Question 4.
Explain the mechanism of dehydration of alcohol.
Answer:
Dehydration of alcohol :
(i) When ethyl alcohol is heated in excess of cone. H2SO4 molecule of water is eliminated and alkene is formed.
![]()
Mechanism :
(i) Protonation of alcohol by H2SO4

(ii) Removal of water

(iii) Elimination of β – hydrogen in the form of proton by base (bisulphate ion)
![]()
Stability of the carbocation (I) determines the case of dehydration and order of stability of carbocation is:
CH3 < C2H5 < Isopropyl < Tertiary butyl
Question 5.
How is ethyl alcohol obtained by molasses? Explain in brief.
Or,
What are molasses? How is alcohol obtained by fermentation? Explain. Tell favourable conditions of fermentation. Draw labelled diagram of coffee still.
Answer:
From molasses:
Molasses is the syrupy solution of sugar left after the separation of cane sugar or beet sugar crystals from the concentrated juice.
The different steps of the manufacture processes are :
1. Dilution:
The molasses is diluted with water so that a concentration of 8 – 10 percent sugar is obtained in solution. This is acidified with dilute sulphuric acid to retard other bacterial growth. A solution of ammonium salts is also added which acts as food for the ferment.
2. Alcoholic fermentation:
The dilute solution obtained above [From step (a)] is taken in big fermentation tanks and some yeast is added. The mixture is kept for a few days and the temperature is maintained at about 30°C. The fermentation reaction starts and the enzyme Invertase (From Yeast) converts sucrose into glucose and fructose which are then converted into

The fermentation is completed in about 3 days. The carbon dioxide is collected as a by product.
3. Distillation:
The fermented liquor is technically called wash or wort which contains about 9 – 10 percent ethanol. It is then distilled in a continuous still called Coffey’s still. It consists of two tall fractionating columns which are called analyser and the rectifier. It works on the counter current principle and the steam and wash travel in opposite directions through the still.

The steam goes upwards in the analyser and takes away the alcohol vapours from the downcoming dilute alcohol. The mixture leaves the analyser from the top and enters the rectifier at the base. Here it heats the wash flowing through the pipes on its way to the analyser. Most of the steam condenses and the alcohol vapours condenses in the condenser. The distillate contain 90% alcohol.
4. Rectification : Wash is rectified by fractional distillation.
![]()
Question 6.
Give equations for three methods of preparation of phenol.
Answer:
Methods of preparation of phenol:
1. By the hydrolysis of Benzene diazonium salts:
Benzene diazonium salt is formed by aromatic primary amine (aniline) with nitrous acid at 0 – 5°C. On boiling aqueous solution of this salt phenol is formed.

2. By alkaline fusion of sodium benzene sulphonate:
On fusing sodium benzene sulphonate with NaOH, sodium phenoxide is formed which on acidification forms phenol.

3. Rasching method:
On heating benzene with mixture of HCl and air to 230°C in the presence of Cu catalyst chlorobenzene is formed which on hydrolysis form phenol.

Question 7.
Write IUPAC names of the following compounds: (NCERT)

Answer:
- 2,2,4 – Trimethyl pentan – 3 – ol
- 5 – Ethylheptane – 2,4 – diol
- Butan – 2,3 – diol
- Propane – 1,2,3 – triol
- 2 – Methylphenol
- 4 – Methylphenol
- 2,5 – Dimethylphenol
- 2,6 – Dimethylphenol
- 1 – Methoxy – 2 – methylpropane
- Ethoxybenzene
- 1 – Phenoxyheptane
- 2 – Ethoxybutane.
Question 8.
How can you obtained following compounds from phenol: (MP 2012; Supp. 14,16)
- 4, 6 – Tribromophenol
- Picric acid
- Aniline
- Benzene
- Phenolp – hthalene
- p – cresol, o – cresol.
Answer:
1. Phenol to Tribromophenol : Phenols readily react with halogens to give polyhalogen substituted compounds. Phenol gives white precipitate of 2, 4, 6 – tribromo – phenol with bromine water.

2. Phenol to Picric acid : Nitration : On nitration, phenols give a variety of products depending upon the conditions.

Nitration of phenol with cone. HNO3 in presence of cone. H2SO4 gives, 2,4,6 – Trini – trophenol (Picric acid). (MP 2014)

3. Phenol to Aniline:

4. Phenol to Benzene :
![]()
5. Phenol to Phenolphthalene : Phenol condenses with phthalic anhydride in presence of cone. H2SO4 to give phenolphthalein which is an indicator for acid – base titrations and is used as a laxative in medicine.

(vi) Phenol to para cresol :
